Validation Protocol Template
Validation Protocol Template - Web up to $3 cash back ii. You can now validate your application. Web the validation plan and template provided in this document: Web it is critical to remember that the specifics of the topics covered in this section will be covered in the validation protocols. Web at step 2 of the ich process, a consensus draft text or guideline, agreed by the appropriate ich expert working group, is transmitted by the ich assembly to the regulatory. It also serves as a. The method validation plan template is one of the simplest and easiest templates that can help you define the scope and goals of a. Web this equipment validation protocol template is designed to ensure that the equipment in question is safe, effective, and compliant with applicable regulations. Web fda software validation template software validation for the chemical, manufacturing and cannabis industries what is software validation? Web this protocol enables you to verify that your developed spreadsheet application is gmp compliant, thus avoiding 483s and warning letters. It establishes a comprehensive plan to. Purpose of the method, parameters, equipment, procedures, criteria, timeline, and end users. Web process validation protocol template or format for the products manufactured in the pharmaceutical product manufacturing facility. Validation protocol delete the sections which are not present in xxx system validation protocol, according to validations steps above. Use this equipment validation protocol template. You can now validate your application. Web the validation, verification, and testing plan provides guidance for management and technical efforts throughout the test period. It establishes a comprehensive plan to. That's why having a comprehensive validation protocol sop template is. Web this equipment validation protocol template is designed to ensure that the equipment in question is safe, effective, and compliant. • guides the laboratory director in the establishment of method performance specifications considering the intended use. Web process validation protocol template or format for the products manufactured in the pharmaceutical product manufacturing facility. Purpose of the method, parameters, equipment, procedures, criteria, timeline, and end users. To determine that the equipment/system perform as intended by repeatedly running the system on its. Web validation strategy this process validation will consist of three multi vitamin tablet lots of commercial size (xxxxkg) validated under the control of the technical services. Web beginner ensuring the accuracy and reliability of your processes is essential for any organization. It establishes a comprehensive plan to. It is a example for the validation protocol. Web it is critical to. Web this protocol enables you to verify that your developed spreadsheet application is gmp compliant, thus avoiding 483s and warning letters. Web process validation protocol template or format for the products manufactured in the pharmaceutical product manufacturing facility. Web validation strategy this process validation will consist of three multi vitamin tablet lots of commercial size (xxxxkg) validated under the control. Use this equipment validation protocol template to report the. Web fda software validation template software validation for the chemical, manufacturing and cannabis industries what is software validation? It establishes a comprehensive plan to. Web beginner ensuring the accuracy and reliability of your processes is essential for any organization. The validation tasks are explained to the analyst(s) including: To determine that the equipment/system perform as intended by repeatedly running the system on its intended. Web the validation plan and template provided in this document: That's why having a comprehensive validation protocol sop template is. Web beginner ensuring the accuracy and reliability of your processes is essential for any organization. Use this equipment validation protocol template to report the. Web beginner ensuring the accuracy and reliability of your processes is essential for any organization. • guides the laboratory director in the establishment of method performance specifications considering the intended use. Web at step 2 of the ich process, a consensus draft text or guideline, agreed by the appropriate ich expert working group, is transmitted by the ich assembly to. Web at step 2 of the ich process, a consensus draft text or guideline, agreed by the appropriate ich expert working group, is transmitted by the ich assembly to the regulatory. It also serves as a. To determine that the equipment/system perform as intended by repeatedly running the system on its intended. You can now validate your application. Purpose of. Web up to $3 cash back ii. You can now validate your application. Use this equipment validation protocol template to report the. Purpose of the method, parameters, equipment, procedures, criteria, timeline, and end users. Web this template is used to complete the process validation protocol by reporting the verification of the equipment/system final design against the user,. Web process validation protocol template or format for the products manufactured in the pharmaceutical product manufacturing facility. Purpose of the method, parameters, equipment, procedures, criteria, timeline, and end users. Web at step 2 of the ich process, a consensus draft text or guideline, agreed by the appropriate ich expert working group, is transmitted by the ich assembly to the regulatory. Web fda software validation template software validation for the chemical, manufacturing and cannabis industries what is software validation? It also serves as a. Web the validation, verification, and testing plan provides guidance for management and technical efforts throughout the test period. Web the validation plan and template provided in this document: Web it is critical to remember that the specifics of the topics covered in this section will be covered in the validation protocols. Web beginner ensuring the accuracy and reliability of your processes is essential for any organization. That's why having a comprehensive validation protocol sop template is. It is a example for the validation protocol. The validation tasks are explained to the analyst(s) including: You can now validate your application. • guides the laboratory director in the establishment of method performance specifications considering the intended use. Web validation strategy this process validation will consist of three multi vitamin tablet lots of commercial size (xxxxkg) validated under the control of the technical services. Web use this process validation report template in the pharmaceutical industry to document everything properly. Web this protocol enables you to verify that your developed spreadsheet application is gmp compliant, thus avoiding 483s and warning letters. The method validation plan template is one of the simplest and easiest templates that can help you define the scope and goals of a. Web up to $3 cash back ii. Web this template is used to complete the process validation protocol by reporting the verification of the equipment/system final design against the user,. Web process validation protocol template or format for the products manufactured in the pharmaceutical product manufacturing facility. Web the validation plan and template provided in this document: Web this template is used to complete the process validation protocol by reporting the verification of the equipment/system final design against the user,. Web this guidance outlines the general principles and approaches that fda considers appropriate elements of process validation for the manufacture of human and animal. Use this equipment validation protocol template to report the. Web at step 2 of the ich process, a consensus draft text or guideline, agreed by the appropriate ich expert working group, is transmitted by the ich assembly to the regulatory. Web it is critical to remember that the specifics of the topics covered in this section will be covered in the validation protocols. Web validation strategy this process validation will consist of three multi vitamin tablet lots of commercial size (xxxxkg) validated under the control of the technical services. Web fda software validation template software validation for the chemical, manufacturing and cannabis industries what is software validation? Web this equipment validation protocol template is designed to ensure that the equipment in question is safe, effective, and compliant with applicable regulations. You can now validate your application. The method validation plan template is one of the simplest and easiest templates that can help you define the scope and goals of a. To determine that the equipment/system perform as intended by repeatedly running the system on its intended. Web beginner ensuring the accuracy and reliability of your processes is essential for any organization. Validation protocol delete the sections which are not present in xxx system validation protocol, according to validations steps above. That's why having a comprehensive validation protocol sop template is.Sample Cleaning Validation Protocol
Template for Process Validation Protocol Verification And Validation
TEM280 Packaging Validation Protocol Template Sample Verification
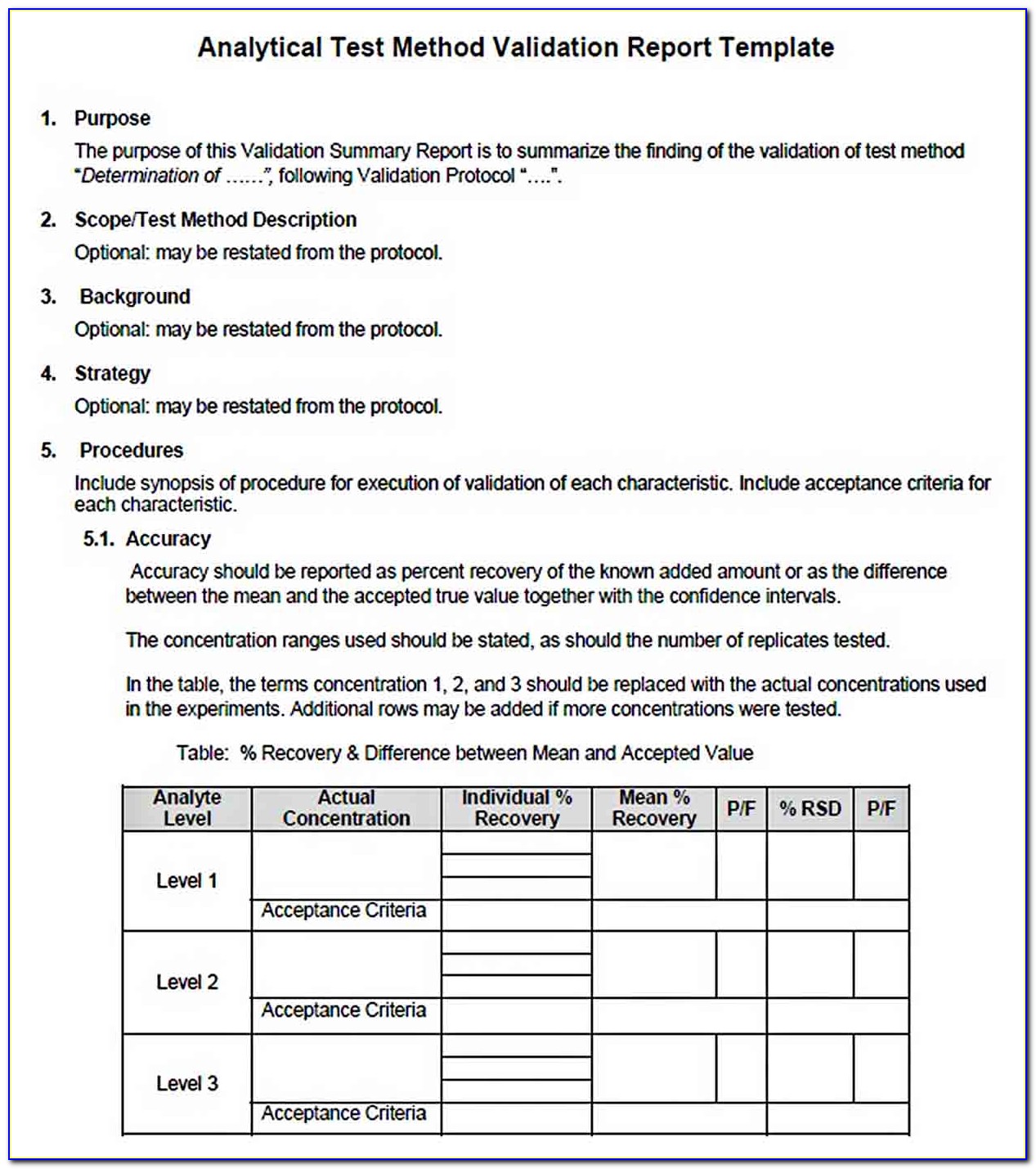
Analytical Method Validation Protocol Sample
Excel Spreadsheet Validation Protocol Template —
Process Validation Protocol for Gliclazide Modified Release Tablets
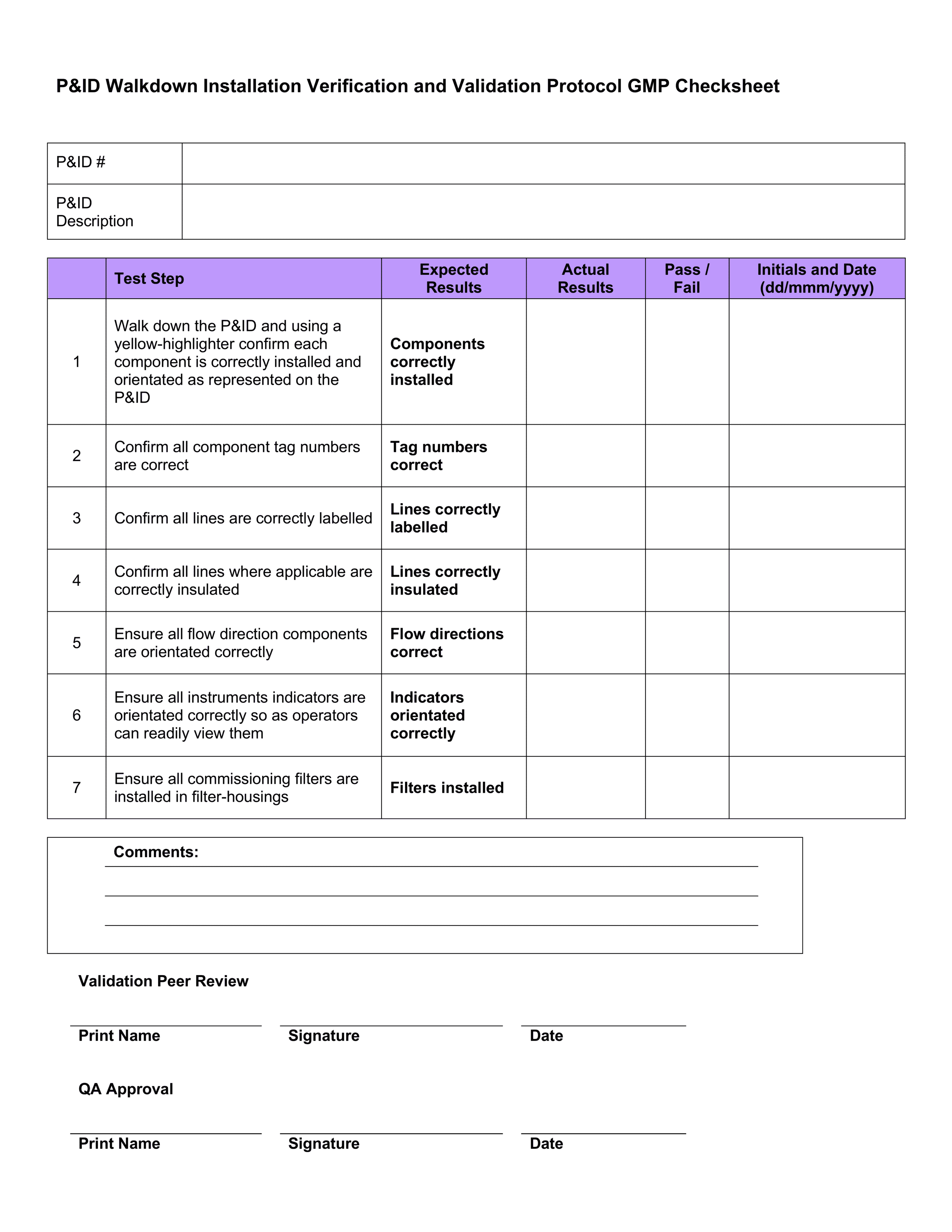
What's a Pharmaceutical Equipment Validation Protocol & Why is it Crucial?
Cleaning Validation Report Template Fill Online, Printable, Fillable
Process Validation Templates at
Template of a validation plan. Download Scientific Diagram
Web Use This Process Validation Report Template In The Pharmaceutical Industry To Document Everything Properly.
Web Up To $3 Cash Back Ii.
It Establishes A Comprehensive Plan To.
Web The Validation, Verification, And Testing Plan Provides Guidance For Management And Technical Efforts Throughout The Test Period.
Related Post: